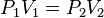You and BoulderJohn are correct. The relevant equation is P1V1 = P2V2. For a free diver taking a breath at the surface and holding it throughout the dive the change in lung volume can be deduced from V2 = P1V1/P2. The ambient pressure of the water, P2, is higher than the surface pressure so with P1V1 being constant V2, the new lung volume at depth, must decrease. The original pressure P1 is constant - you are not adding or removing air, and the original volume is constant, i.e. at the start of the dive the volume is not changing.
Keep in mind that the amount of on and offgassing is only dependent on the pp (partial pressure) of N2. Lung volume is determined by the total pressure or the sum of all the pp's of each gas. In another thread I mentioned that both O2 and CO2 exchange across the lung-blood barrier in different directions at the same time, O2 at a much higher pressure drop than CO2. The flow of any gas across this barrier is by diffusion which is fundamentally different than by the force of a pressure difference as would be the case for an embolism. However, a change of pp will effect the total pressure since the pp's are additive. In regards to N2 on/offgassing for a free diver on surface air you might be thinking of total lung pressure increasing with ambient pressure thereby "forcing" more N2 to the tissues. But, the pressure in the lungs is constant and with it the pp's of each constituent gas. Therefore, as the ambient pressure increases the assoiated rise in pressure drop will cause a decrease in volume because the lungs are a flexible container and the diver is not adding or venting gas. (Sorry for the long explanation but I hope this is clearer.