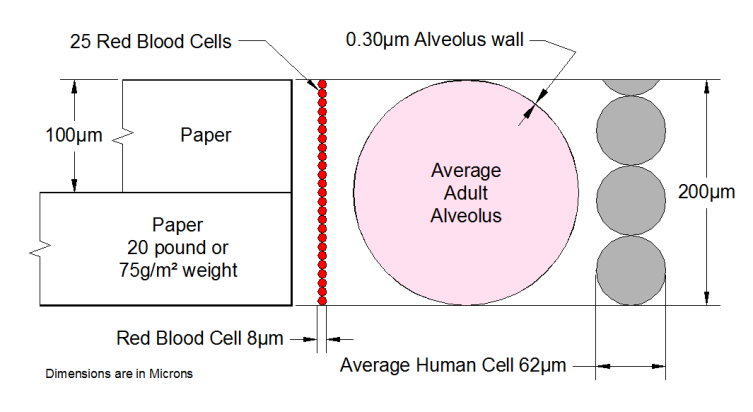
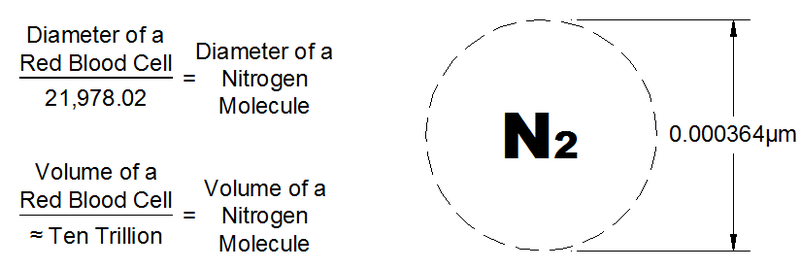
May I then respectfully suggest that you don't make definitive statements about things you are recalling, perhaps erroneously, from long ago? At least put in a disclaimer or two? Because some of the things you have said in this thread have been plain wrong.
Covalent bond strength isn't comparable to ionic lattice energy (
cite). The average bond enthalpy of of the C-H bond in methane is 415 kJ/mol, and the bond enthalpy of the H-H bond in H2 is 436 kJ/mol (ibid.) The lattice enthalpy of NaCl is 787 kJ/mol (
cite). So why does NaCl dissolve in water as free, dissociated ions? Because the lattice enthalpy is the enthalpy change when going from solid salt to gaseous ions, and that's not what happens here. You have to consider the end state, which is sodium and chloride ions in water. So the net enthalpy change is only 3.9 kJ/mol. Which still is unfavorable, but we're just comparing enthalpies here. The other important driver in chemical reactions is entropy. Look at a standard ice pack. The dissolution of urea or ammonium nitrate in water is highly endothermic, IOW if we just looked at the enthalpy change, it would never happen. But here comes entropy and drives the dissolution anyway, and the enthalpy change is provided by the cooling of the solution.
Tl,dr: you can't just compare "bond strength" of covalent bonds and ionic bonds to say which is the "strongest". You have to consider the whole system, both starting point and end state.
And yes, I have a chemistry degree. In case someone was still wondering about that. And thermodynamics is actually quite interesting, because that's what makes everything work. Literally everything.